Water is the universal solvent due to its unique ability to dissolve a wide array of substances, vital for life on Earth. At WHY.EDU.VN, we delve into water’s solvency, exploring its chemical composition and the physical attributes that enable it to transport essential chemicals, minerals, and nutrients through air, ground, and our bodies, thus making it the universal dissolver. Discover how this polarity helps dissolve compounds like salt and supports bodily functions, ensuring health and well-being.
1. Understanding Water’s Universal Solvent Properties
Water is frequently referred to as the “universal solvent” because it can dissolve more substances than any other liquid. This property is essential for all living things on Earth. Water’s role as a solvent enables it to carry valuable chemicals, minerals, and nutrients through the air, ground, and our bodies. This makes water indispensable for sustaining life and various environmental processes.
1.1. Polarity and Chemical Composition
Water’s unique chemical composition and physical attributes make it such an excellent solvent. Water molecules have a polar arrangement of oxygen and hydrogen atoms. One side (hydrogen) has a positive electrical charge, and the other side (oxygen) has a negative charge. This polarity allows water molecules to be attracted to many different types of molecules.
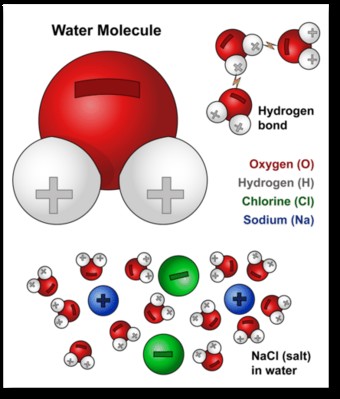 Water molecule polarity showing positive and negative charges
Water molecule polarity showing positive and negative charges
1.2. How Water Dissolves Substances
Water can become so heavily attracted to a different compound, like salt (NaCl), that it can disrupt the attractive forces that hold the sodium and chloride in the salt compound together, thus dissolving it. This process is critical in various biological and environmental systems.
1.3. Significance in Biological Systems
In biological systems, water’s solvent properties are crucial for transporting nutrients and removing waste. For example, our kidneys rely on water to dissolve and filter out substances from the foods and drinks we consume, ensuring that these substances are eliminated from our bodies efficiently.
2. Detailed Look at Water’s Molecular Interactions
To understand why water is the universal solvent, it’s essential to examine its molecular interactions with other substances. Water’s polarity allows it to form strong attractions with ions and other polar molecules, facilitating dissolution.
2.1. Electrical Charges and Molecular Bonds
At the molecular level, salt dissolves in water because both water and salt compounds are polar, with positive and negative charges on opposite sides in the molecule. The bonds in salt compounds are called ionic because they both have an electrical charge—the chloride ion is negatively charged, and the sodium ion is positively charged.
2.2. Covalent Bonds in Water Molecules
A water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge. When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules.
2.3. The Dissolution Process Step-by-Step
- Attraction: The positively charged side of the water molecules are attracted to the negatively charged chloride ions, and the negatively charged side of the water molecules are attracted to the positively charged sodium ions.
- Disruption: Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.
- Surrounding: After the salt compounds are pulled apart, the sodium and chloride atoms are surrounded by water molecules.
- Homogeneous Solution: Once this happens, the salt is dissolved, resulting in a homogeneous solution.
3. Water’s Role in Human Physiology
Water plays a vital role in human physiology, supporting numerous bodily functions, including digestion, nutrient transport, and waste removal. Its solvent properties are particularly important in these processes.
3.1. Kidneys and Waste Removal
Our kidneys and water’s solvent properties make a great pair in keeping us alive and healthy. The kidneys are responsible for filtering out substances that enter our bodies from the foods and drinks we consume. These substances need to be eliminated after they accumulate, and that is where water helps out.
3.2. Water as a Solvent in the Kidneys
Being such a great solvent, water washing through the kidneys dissolves these substances and sends them on the way out of our bodies. This process is essential for maintaining electrolyte balance and removing toxic compounds.
3.3. Other Physiological Processes
In addition to waste removal, water also aids in nutrient absorption in the intestines and the transport of these nutrients throughout the body via the bloodstream. Its solvency ensures that these essential compounds are readily available to cells.
4. Environmental Significance of Water as a Solvent
Water’s solvent properties are not only important in biological systems but also in various environmental processes. It plays a crucial role in weathering, erosion, and the transport of pollutants.
4.1. Weathering and Erosion
Water’s ability to dissolve minerals and rocks contributes to weathering and erosion. Over time, water can break down rocks and transport dissolved minerals, shaping landscapes and influencing soil composition.
4.2. Transport of Nutrients and Pollutants
In aquatic ecosystems, water acts as a medium for transporting nutrients and pollutants. Dissolved nutrients support the growth of aquatic plants and animals, while dissolved pollutants can have detrimental effects on water quality and aquatic life.
4.3. Impact on Water Quality
Water’s solvent properties also affect water quality. Dissolved minerals and chemicals can alter the pH, hardness, and taste of water, affecting its suitability for drinking and other uses.
5. Comparative Analysis: Water vs. Other Solvents
While water is often called the universal solvent, it is not the only solvent. Other liquids, such as organic solvents, can also dissolve substances, but they have different properties and applications.
5.1. Polarity Comparison
One key difference between water and other solvents is polarity. Water is a polar solvent, meaning it can dissolve polar and ionic compounds more effectively than nonpolar compounds. Nonpolar solvents, such as oil, can dissolve nonpolar substances like fats and oils.
5.2. Applications of Different Solvents
The choice of solvent depends on the substance being dissolved and the application. Water is commonly used in biological and environmental applications, while organic solvents are used in industrial processes and chemical reactions.
5.3. Limitations of Water as a Solvent
Despite its versatility, water has limitations as a solvent. It cannot dissolve nonpolar substances effectively, and it can react with certain compounds. In these cases, other solvents may be more suitable.
6. Factors Affecting Water’s Solvent Capability
Several factors can affect water’s ability to dissolve substances, including temperature, pressure, and the presence of other solutes. Understanding these factors is essential for optimizing water’s use as a solvent.
6.1. Temperature Effects
Temperature can significantly affect water’s solvent capability. In general, the solubility of solids and liquids in water increases with temperature, while the solubility of gases decreases.
6.2. Pressure Effects
Pressure can also affect water’s solvent capability, particularly for gases. Higher pressure increases the solubility of gases in water.
6.3. Presence of Other Solutes
The presence of other solutes can affect water’s solvent capability. For example, the solubility of a salt in water may decrease if other salts are already dissolved in the water. This is known as the common ion effect.
7. Practical Applications of Water’s Solvent Properties
Water’s solvent properties are utilized in a wide range of practical applications, from household cleaning to industrial processes.
7.1. Household Cleaning
Water is commonly used as a solvent in household cleaning products. It can dissolve dirt, grime, and other substances, making it an effective cleaning agent.
7.2. Industrial Processes
In industrial processes, water is used as a solvent for chemical reactions, extraction, and purification. Its ability to dissolve a wide range of substances makes it a versatile solvent for various applications.
7.3. Pharmaceutical Applications
Water is also used as a solvent in pharmaceutical applications. It can dissolve drugs and other substances, making it easier to administer them to patients.
8. Research and Studies on Water’s Solvent Abilities
Numerous research studies have investigated water’s solvent abilities, providing valuable insights into its molecular interactions and practical applications.
8.1. Molecular Dynamics Simulations
Molecular dynamics simulations have been used to study the interactions between water molecules and other substances at the atomic level. These simulations can provide insights into the mechanisms of dissolution and the factors that affect water’s solvent capability.
8.2. Spectroscopic Studies
Spectroscopic studies have been used to investigate the structure and dynamics of water molecules in solution. These studies can provide information about the interactions between water and other solutes.
8.3. Environmental Studies
Environmental studies have investigated the role of water as a solvent in the transport of pollutants and nutrients in aquatic ecosystems. These studies can inform strategies for managing water quality and protecting aquatic life.
9. Addressing Common Misconceptions About Water as a Solvent
Despite its importance, there are several common misconceptions about water as a solvent. Addressing these misconceptions can help people better understand water’s properties and applications.
9.1. Water Dissolves Everything
One common misconception is that water can dissolve everything. While water can dissolve a wide range of substances, it cannot dissolve nonpolar substances effectively.
9.2. Pure Water is the Best Solvent
Another misconception is that pure water is the best solvent. In some cases, the presence of other solutes can enhance water’s solvent capability.
9.3. Water is Always Safe
Finally, some people believe that water is always safe. However, water can contain dissolved pollutants that can be harmful to human health.
10. Future Directions in Water Solvent Research
Research on water as a solvent is ongoing, with future directions focusing on improving our understanding of its molecular interactions and developing new applications.
10.1. Nanotechnology Applications
Nanotechnology is being used to develop new materials that can enhance water’s solvent capability. These materials can be used in water treatment, drug delivery, and other applications.
10.2. Sustainable Solvent Development
Researchers are also working on developing sustainable solvents that can replace traditional organic solvents. Water is a promising candidate for a sustainable solvent due to its abundance and low toxicity.
10.3. Climate Change Implications
Climate change is affecting water availability and quality around the world. Research is needed to understand how these changes will affect water’s solvent capability and its role in environmental processes.
11. The Science Behind Why Water Is Called the Universal Solvent
Water’s exceptional solvent capabilities are deeply rooted in its molecular structure, a subject of continuous exploration by scientists worldwide. This section delves into the intricacies of water’s unique properties that lead to its universal solvent status, referencing key scientific findings and ongoing research.
11.1. The Chemistry of Water: A Polar Molecule
Water (H2O) is a polar molecule, which means it has a slightly positive charge on one side (hydrogen atoms) and a slightly negative charge on the other (oxygen atom). This polarity arises from the unequal sharing of electrons between the oxygen and hydrogen atoms. Oxygen is more electronegative than hydrogen, pulling the electrons closer and creating a partial negative charge (δ-) on the oxygen atom and partial positive charges (δ+) on the hydrogen atoms.
11.2. Hydrogen Bonding: The Key to Water’s Cohesion and Adhesion
The polarity of water molecules allows them to form hydrogen bonds with each other. Hydrogen bonding occurs when the partially positive hydrogen atom of one water molecule is attracted to the partially negative oxygen atom of another. These bonds are relatively weak compared to covalent bonds but are strong enough to create significant cohesion (attraction between like molecules) and adhesion (attraction between unlike molecules).
Caption: Illustration of hydrogen bonds between water molecules, facilitating cohesion and adhesion.
11.3. Water’s Ability to Dissolve Ionic Compounds
Ionic compounds, such as sodium chloride (NaCl), are composed of ions with positive (cations) and negative (anions) charges. When an ionic compound is placed in water, the polar water molecules surround the ions. The partially negative oxygen atoms are attracted to the positive sodium ions (Na+), and the partially positive hydrogen atoms are attracted to the negative chloride ions (Cl-).
11.4. Water’s Role in Dissolving Polar Covalent Compounds
Polar covalent compounds also dissolve in water due to the interaction between their polar regions and water molecules. For example, ethanol (C2H5OH) is a polar molecule because the oxygen atom is more electronegative than the carbon and hydrogen atoms. When ethanol is mixed with water, hydrogen bonds form between the partially negative oxygen atom of ethanol and the partially positive hydrogen atoms of water, and vice versa. These interactions stabilize the ethanol molecules in the water solution, allowing ethanol to dissolve.
11.5. Limitations of Water as a Solvent for Nonpolar Compounds
Nonpolar compounds, such as oils and fats, do not dissolve well in water. This is because nonpolar molecules do not have partially charged regions that can interact with the polar water molecules. The nonpolar molecules tend to cluster together, minimizing their contact with water. This phenomenon is known as hydrophobic interaction, and it is driven by the tendency of water to maximize its hydrogen bonding with other water molecules.
12. Water’s Role in Various Industries
Water’s solvent properties are indispensable in various industries, influencing processes ranging from pharmaceutical manufacturing to food processing. This segment highlights the critical roles water plays and underscores its economic and functional significance.
12.1. Water in the Pharmaceutical Industry
Water is a critical component in the pharmaceutical industry, serving as a solvent, reactant, and cleaning agent. Its solvent properties are used to dissolve active pharmaceutical ingredients (APIs) and excipients, ensuring uniform dosage and effective drug delivery.
12.2. Water’s Utility in the Food and Beverage Industry
In the food and beverage industry, water is used extensively as a solvent, cleaning agent, and ingredient. It is used to dissolve sugars, salts, acids, and other ingredients in food and beverage products.
12.3. Water in the Chemical Industry
The chemical industry relies heavily on water as a solvent, coolant, and reactant. It is used in a wide range of chemical processes, including the production of acids, bases, salts, and organic chemicals. Water’s ability to dissolve a wide range of substances makes it an ideal solvent for many chemical reactions.
12.4. Water in the Energy Sector
Water plays a crucial role in the energy sector, particularly in power generation. It is used as a coolant in thermal power plants (coal, nuclear, and natural gas) to absorb heat and produce steam, which drives turbines to generate electricity. Water’s high heat capacity and thermal conductivity make it an excellent coolant.
12.5. Water Use in Agriculture
Agriculture is one of the largest consumers of water globally. Water is used for irrigation to support crop growth and is essential for dissolving and transporting nutrients from the soil to plants.
13. Environmental Impact of Water Pollution
Water pollution poses significant environmental challenges, affecting ecosystems and human health. This section covers the sources, effects, and potential solutions for mitigating water pollution.
13.1. Sources of Water Pollution
Water pollution can come from various sources, including industrial discharge, agricultural runoff, sewage, and oil spills. These sources introduce pollutants such as heavy metals, pesticides, fertilizers, organic matter, and pathogens into water bodies.
13.2. Environmental Effects of Water Pollution
Water pollution can have detrimental effects on aquatic ecosystems, impacting biodiversity and ecosystem functions. Pollutants can kill aquatic organisms, disrupt food chains, and reduce the overall health of aquatic environments.
13.3. Human Health Impacts of Water Pollution
Contaminated water can transmit diseases such as cholera, typhoid fever, dysentery, and hepatitis A. Exposure to chemical pollutants in drinking water can lead to chronic health problems, including cancer, reproductive disorders, and neurological damage.
13.4. Mitigation Strategies for Water Pollution
Mitigating water pollution requires a multi-faceted approach that includes regulations, technological solutions, and public awareness. Implementing stricter environmental regulations can help control industrial discharge and agricultural runoff.
13.5. The Role of Water Treatment Technologies
Water treatment technologies play a crucial role in removing pollutants from water sources, ensuring safe drinking water and protecting aquatic ecosystems. Conventional water treatment processes include coagulation, flocculation, sedimentation, filtration, and disinfection.
14. Water Treatment Processes and Technologies
Water treatment is essential for ensuring the availability of safe and clean water for various uses. This section outlines the processes and technologies used to treat water for drinking, industrial, and environmental purposes.
14.1. Conventional Water Treatment Processes
Conventional water treatment processes typically involve several steps to remove contaminants from raw water sources. These steps include:
- Coagulation and Flocculation: Chemicals are added to the water to cause small particles to clump together, forming larger, heavier particles called flocs.
- Sedimentation: The water is allowed to sit quietly, allowing the flocs to settle to the bottom of the tank.
- Filtration: The water is passed through filters, such as sand or activated carbon, to remove any remaining particles.
- Disinfection: The water is treated with chlorine, ozone, or ultraviolet (UV) light to kill any remaining pathogens.
14.2. Advanced Water Treatment Technologies
In addition to conventional processes, advanced water treatment technologies are used to remove specific contaminants or to improve the overall quality of treated water. These technologies include:
- Membrane Filtration: Processes such as reverse osmosis (RO) and ultrafiltration (UF) use membranes to filter out contaminants from water.
- Activated Carbon Adsorption: Activated carbon is used to remove organic compounds, taste, and odor from water.
- Ion Exchange: Ion exchange resins are used to remove specific ions, such as nitrates or perchlorates, from water.
- Advanced Oxidation Processes (AOPs): AOPs use oxidants such as ozone, hydrogen peroxide, and UV light to degrade organic pollutants in water.
14.3. Wastewater Treatment Processes
Wastewater treatment is the process of removing contaminants from domestic and industrial wastewater. The goals of wastewater treatment are to protect public health, prevent water pollution, and conserve water resources.
14.4. Decentralized Water Treatment Systems
Decentralized water treatment systems are small-scale treatment facilities that are located close to the source of wastewater or drinking water demand. These systems can be used to treat wastewater from individual homes, businesses, or communities.
14.5. Future Trends in Water Treatment
The future of water treatment is likely to be driven by several factors, including increasing water scarcity, stricter environmental regulations, and advancements in technology.
15. Water Conservation and Sustainability
Water conservation and sustainability are crucial for ensuring the availability of water resources for future generations. This section outlines strategies for conserving water in various sectors and promoting sustainable water management practices.
15.1. Water Conservation in Agriculture
Agriculture is the largest consumer of water globally, accounting for about 70% of total water withdrawals. Implementing water-efficient irrigation technologies, such as drip irrigation and micro-sprinklers, can significantly reduce water use in agriculture.
15.2. Industrial Water Conservation Strategies
Industries can implement various strategies to reduce water consumption, including:
- Water Audits: Conducting water audits to identify areas where water is being wasted.
- Water Recycling: Recycling water used in industrial processes for other applications.
- Process Optimization: Optimizing industrial processes to reduce water consumption.
- Water-Efficient Equipment: Installing water-efficient equipment and fixtures.
15.3. Residential Water Conservation Tips
Households can conserve water by adopting simple water-saving practices, such as:
- Fixing Leaks: Repairing leaky faucets and toilets.
- Water-Efficient Appliances: Using water-efficient appliances, such as washing machines and dishwashers.
- Shorter Showers: Taking shorter showers.
- Watering Wisely: Watering lawns and gardens efficiently, preferably during the early morning or late evening to minimize evaporation.
15.4. Sustainable Water Management Practices
Sustainable water management involves using water resources in a way that meets the needs of the present without compromising the ability of future generations to meet their own needs. This requires a holistic approach that considers the environmental, social, and economic aspects of water use.
15.5. The Role of Policy and Governance
Effective water management requires strong policy and governance frameworks. Governments can play a crucial role in promoting water conservation and sustainability through regulations, incentives, and public awareness campaigns.
16. The Importance of Water Quality Monitoring
Water quality monitoring is essential for assessing the health of water bodies and ensuring the safety of drinking water. This section covers the parameters used to assess water quality and the technologies used for water quality monitoring.
16.1. Key Water Quality Parameters
Several parameters are used to assess water quality, including:
- pH: A measure of the acidity or alkalinity of water.
- Temperature: Water temperature can affect the solubility of pollutants and the metabolism of aquatic organisms.
- Dissolved Oxygen (DO): The amount of oxygen dissolved in water, which is essential for aquatic life.
- Turbidity: A measure of the cloudiness of water, which can affect light penetration and the growth of aquatic plants.
- Nutrients: The concentration of nutrients such as nitrogen and phosphorus, which can contribute to eutrophication.
- Pathogens: The presence of disease-causing microorganisms, such as bacteria and viruses.
- Chemical Contaminants: The concentration of chemical pollutants, such as heavy metals, pesticides, and organic compounds.
16.2. Water Quality Monitoring Technologies
Water quality monitoring can be conducted using a variety of technologies, including:
- Field Measurements: Using portable instruments to measure water quality parameters in the field.
- Laboratory Analysis: Collecting water samples and analyzing them in a laboratory.
- Remote Sensing: Using satellites and drones to monitor water quality over large areas.
- Real-Time Monitoring Systems: Deploying sensors in water bodies to continuously monitor water quality parameters and transmit data to a central location.
16.3. The Role of Citizen Science
Citizen science initiatives engage volunteers in water quality monitoring, providing valuable data and raising public awareness about water quality issues. Citizen scientists can collect water samples, measure water quality parameters, and report their findings to environmental agencies.
16.4. Data Management and Analysis
Effective water quality monitoring requires robust data management and analysis systems. Water quality data should be stored in a centralized database and analyzed to identify trends, assess the effectiveness of pollution control measures, and inform water management decisions.
16.5. The Importance of Water Quality Standards
Water quality standards establish the maximum allowable concentrations of pollutants in water bodies. These standards are used to protect public health, aquatic life, and other beneficial uses of water.
17. Case Studies of Water’s Role as a Solvent
Examining specific examples underscores the importance of water’s solvent properties across diverse scenarios, reinforcing its status as the universal solvent.
17.1. Water’s Role in Karst Topography Formation
Karst topography is formed by the dissolution of soluble rocks such as limestone, dolomite, and gypsum. Rainwater, which is slightly acidic due to the absorption of carbon dioxide from the atmosphere, dissolves these rocks over time, creating underground drainage systems, caves, sinkholes, and other distinctive features.
17.2. Water as a Solvent in Geothermal Systems
Geothermal systems harness heat from the Earth’s interior to generate electricity and heat buildings. Water acts as a heat transfer fluid in geothermal systems, circulating through underground reservoirs and absorbing heat from the surrounding rocks.
17.3. Water in the Extraction of Minerals
Water is used as a solvent in the extraction of minerals from ores. For example, in the mining of gold, cyanide solutions are used to dissolve gold from the ore, forming a gold-cyanide complex that can be recovered through chemical processes.
17.4. Water’s Role in the Haber-Bosch Process
The Haber-Bosch process is an industrial process for producing ammonia from nitrogen and hydrogen. Ammonia is a key ingredient in fertilizers and is essential for modern agriculture. Water is used to produce hydrogen through steam reforming of natural gas.
17.5. Water in the Production of Paper
Water is used extensively in the production of paper, serving as a solvent, suspending agent, and transport medium. In the pulping process, wood chips are mixed with water and chemicals to break down the lignin and separate the cellulose fibers.
18. Innovations in Water Purification Technologies
Advancements in water purification technologies are continually enhancing our ability to access clean and safe water. This section explores some of the latest innovations.
18.1. Nanomaterials for Water Purification
Nanomaterials, such as carbon nanotubes, graphene, and metal nanoparticles, have shown great potential for water purification. These materials can be used to remove a wide range of contaminants, including heavy metals, bacteria, viruses, and organic pollutants.
18.2. Biofiltration Systems
Biofiltration systems use microorganisms to remove pollutants from water. These systems typically consist of a filter bed containing a layer of sand or gravel, which is colonized by bacteria, fungi, and other microorganisms.
18.3. Solar Water Disinfection (SODIS)
Solar water disinfection (SODIS) is a simple and low-cost method for disinfecting water using sunlight. Water is placed in clear plastic bottles and exposed to direct sunlight for several hours. The UV radiation from the sun kills pathogens in the water, making it safe to drink.
18.4. Forward Osmosis (FO)
Forward osmosis (FO) is a membrane-based separation process that uses osmotic pressure to drive water through a semi-permeable membrane, separating it from contaminants. FO does not require external pressure and can be used to treat a wide range of water sources.
18.5. Capacitive Deionization (CDI)
Capacitive deionization (CDI) is an electrochemical method for removing ions from water. Water is passed between two electrodes that are charged with a low voltage. The ions are attracted to the electrodes, removing them from the water.
19. The Future of Water Resources Management
The future of water resources management will require a more integrated and sustainable approach that considers the environmental, social, and economic aspects of water use.
19.1. Integrated Water Resources Management (IWRM)
Integrated Water Resources Management (IWRM) is a holistic approach to managing water resources that considers the interconnections between different water uses and the environment. IWRM aims to balance competing demands for water and ensure that water resources are used sustainably.
19.2. The Water-Energy Nexus
The water-energy nexus refers to the interdependencies between water and energy. Water is used in the production of energy, and energy is used in the treatment and distribution of water. Managing the water-energy nexus is essential for ensuring the sustainability of both water and energy resources.
19.3. Climate Change Adaptation
Climate change is affecting water availability and quality around the world. Adapting to climate change will require a range of measures, including:
- Water Conservation: Reducing water consumption in all sectors.
- Water Storage: Increasing water storage capacity through dams, reservoirs, and groundwater recharge.
- Drought Management: Developing drought management plans to cope with water shortages.
- Flood Control: Implementing flood control measures to protect communities and infrastructure from floods.
19.4. The Role of Technology
Technology will play a crucial role in the future of water resources management. Advanced technologies can be used to monitor water resources, improve water treatment processes, and reduce water consumption.
19.5. The Importance of Collaboration
Effective water resources management requires collaboration between governments, businesses, communities, and individuals. Working together, we can ensure that water resources are used sustainably and that everyone has access to safe and clean water.
20. Frequently Asked Questions (FAQ) About Water as a Universal Solvent
This section provides answers to common questions about water’s properties as a universal solvent, offering clear and concise explanations.
Q1: Why is water called the universal solvent?
A1: Water is called the universal solvent because it can dissolve more substances than any other liquid due to its polar nature and ability to form hydrogen bonds.
Q2: What makes water a polar molecule?
A2: Water is a polar molecule because the oxygen atom is more electronegative than the hydrogen atoms, creating a partial negative charge on the oxygen and partial positive charges on the hydrogens.
Q3: How does water dissolve ionic compounds?
A3: Water dissolves ionic compounds by surrounding the ions with water molecules, with the partially negative oxygen atoms attracted to positive ions and the partially positive hydrogen atoms attracted to negative ions.
Q4: Can water dissolve everything?
A4: No, water cannot dissolve everything. It primarily dissolves polar and ionic compounds but struggles with nonpolar substances like oils and fats.
Q5: What factors affect water’s solvent capability?
A5: Factors affecting water’s solvent capability include temperature, pressure, and the presence of other solutes.
Q6: Why is water important in human physiology?
A6: Water is crucial for human physiology as it supports digestion, nutrient transport, waste removal, and numerous other bodily functions.
Q7: How does water help in kidney function?
A7: Water helps kidneys filter out waste substances from the body by dissolving them and facilitating their removal.
Q8: What is the environmental significance of water as a solvent?
A8: Water plays a vital role in environmental processes such as weathering, erosion, and the transport of nutrients and pollutants.
Q9: What are some practical applications of water’s solvent properties?
A9: Practical applications include household cleaning, industrial processes, pharmaceutical applications, and agricultural irrigation.
Q10: What are some emerging technologies for water purification?
A10: Emerging technologies include nanomaterials for water purification, biofiltration systems, solar water disinfection (SODIS), forward osmosis (FO), and capacitive deionization (CDI).
Water’s remarkable properties as a solvent are essential for life, industry, and the environment. Understanding these properties and promoting sustainable water management practices are crucial for ensuring a healthy planet and a sustainable future. For more in-depth answers and expert insights, visit WHY.EDU.VN, your go-to resource for comprehensive explanations and answers to your most pressing questions.
Do you have more questions about water’s unique properties? At WHY.EDU.VN, we connect you with experts who can provide detailed answers and insights. Visit us at 101 Curiosity Lane, Answer Town, CA 90210, United States, or reach out via WhatsApp at +1 (213) 555-0101. Explore more and ask your questions at why.edu.vn today and get the answers you need from trusted professionals.
