Why does ice float on water, a question that has intrigued scientists and curious minds alike? At WHY.EDU.VN, we break down complex scientific concepts into easily digestible explanations. Discover the science behind ice’s buoyancy and explore related phenomena, unveiling the science behind this everyday marvel with comprehensive insights and clear explanations. Dive into the fascinating world of water density, hydrogen bonding, and the unique properties that make ice float.
1. Understanding Buoyancy: The Key to Floating
Buoyancy is the force that allows objects to float. When an object is submerged in water, it experiences an upward force that opposes gravity. This upward force, known as the buoyant force, is what determines whether an object floats or sinks. Let’s delve into Archimedes’ principle, a cornerstone of understanding buoyancy.
1.1 Archimedes’ Principle Explained
Archimedes’ principle states that the buoyant force on an object submerged in a fluid is equal to the weight of the fluid displaced by the object. This principle is fundamental to understanding why some objects float while others sink.
The buoyant force (FB) can be calculated as:
FB = ρwater × Vdisplaced × g
Where:
- ρwater is the density of water
- Vdisplaced is the volume of water displaced by the object
- g is the acceleration due to gravity
Alt: Archimedes principle diagram showing buoyant force acting on an object submerged in water, displacing fluid.
1.2 Density: The Deciding Factor
Density plays a crucial role in determining whether an object floats. Density is defined as mass per unit volume (ρ = m/V). An object will float if its density is less than the density of the fluid it is placed in.
- If the object’s density is less than the water’s density, the buoyant force is greater than the gravitational force, and the object floats.
- If the object’s density is greater than the water’s density, the buoyant force is less than the gravitational force, and the object sinks.
2. The Peculiarity of Water: Why Ice Behaves Differently
Water is unique because its solid form, ice, is less dense than its liquid form. This is unusual because, for most substances, the solid form is denser than the liquid form. This unusual property of water is due to hydrogen bonding.
2.1 Hydrogen Bonding in Water Molecules
A water molecule consists of one oxygen atom and two hydrogen atoms (H2O). The oxygen atom is more electronegative than the hydrogen atoms, meaning it attracts electrons more strongly. This creates a slight negative charge (δ-) on the oxygen atom and slight positive charges (δ+) on the hydrogen atoms.
Alt: Water molecule diagram showing partial charges on oxygen and hydrogen atoms, illustrating electronegativity.
These partial charges allow water molecules to form hydrogen bonds with each other. Hydrogen bonds are weak electrostatic attractions between the slightly positive hydrogen atom of one water molecule and the slightly negative oxygen atom of another water molecule.
2.2 The Crystal Lattice Structure of Ice
When water freezes, the hydrogen bonds cause the molecules to arrange themselves into a crystal lattice structure. This structure is more open and less dense than liquid water. The hydrogen bonds force water molecules to form a hexagonal arrangement, leaving more space between the molecules.
Alt: Crystal lattice structure of ice showing hexagonal arrangement and hydrogen bonds, indicating increased spacing.
2.3 Density Comparison: Ice vs. Liquid Water
Due to the crystal lattice structure, ice is about 9% less dense than liquid water. This density difference is why ice floats on water. The lower density means that a given volume of ice weighs less than the same volume of water, allowing it to be buoyed up by the water.
| Property | Liquid Water | Ice |
|---|---|---|
| Density (at 0°C) | 1000 kg/m³ | 917 kg/m³ |
| Molecular Packing | Close | Open |
| Structure | Disordered | Crystalline |
3. Why Most Substances Sink When Solidified
Unlike water, most substances are denser in their solid form than in their liquid form. This is because, in the solid state, the molecules are more tightly packed together. When a substance freezes, the molecules lose kinetic energy and move closer together, forming a more compact structure.
3.1 Molecular Arrangement in Solids and Liquids
In a solid, molecules are held in fixed positions and vibrate around these positions. In a liquid, molecules have more freedom to move around, but they are still relatively close together. When a substance freezes, the molecules arrange themselves in a way that minimizes the space between them, resulting in a higher density.
3.2 Thermal Expansion and Contraction
Most substances contract when they cool and expand when they heat up. This is because the kinetic energy of the molecules changes with temperature. As a substance cools, the molecules slow down and move closer together, causing the substance to contract. As a substance heats up, the molecules speed up and move further apart, causing the substance to expand.
4. The Impact of Pressure on Freezing Water
Pressure can also affect the freezing point and density of water. Applying pressure to water can lower its freezing point. This is because the application of pressure favors the denser phase of water, which is the liquid phase.
4.1 Phase Diagram of Water
The phase diagram of water shows how the state of water (solid, liquid, or gas) depends on temperature and pressure. The diagram reveals that increasing pressure lowers the melting point of ice.
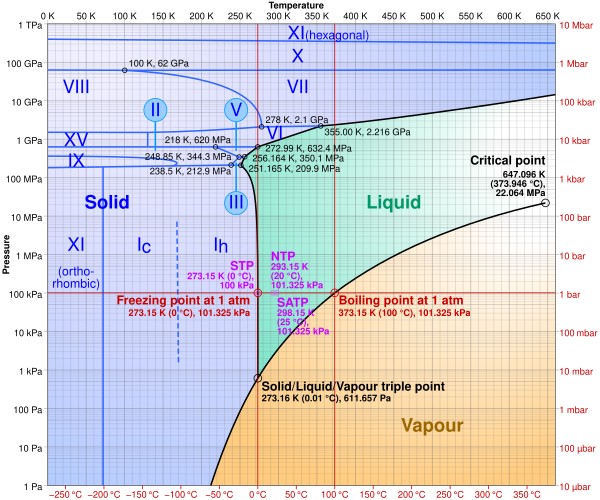 Phase diagram of water illustrating the relationship between temperature, pressure, and the state of water
Phase diagram of water illustrating the relationship between temperature, pressure, and the state of water
Alt: Water phase diagram showing temperature, pressure, and phases of water: solid, liquid, and gas.
4.2 Different Forms of Ice
Under different pressure and temperature conditions, water can form different crystalline structures of ice. There are at least 19 known crystalline forms of ice, each with different densities and properties. Ordinary ice, which we encounter in everyday life, is known as ice Ih.
| Ice Form | Density (kg/m³) | Temperature (°C) | Pressure (MPa) |
|---|---|---|---|
| Ice Ih | 917 | 0 | 0.1 |
| Ice II | 1160 | -35 | 200 |
| Ice III | 1150 | -25 | 300 |
5. Real-World Implications of Ice Floating
The fact that ice floats has significant implications for the environment and life on Earth. If ice were denser than water, it would sink to the bottom of bodies of water, causing them to freeze from the bottom up.
5.1 Insulation for Aquatic Life
Floating ice acts as an insulator for the water below, preventing it from freezing solid. This allows aquatic life to survive during cold winter months. The layer of ice at the surface protects the water underneath from extreme cold, maintaining a stable environment for fish and other organisms.
5.2 Climate Regulation
Ice and snow cover a significant portion of the Earth’s surface, especially in polar regions. This ice reflects a large amount of sunlight back into space, helping to regulate the Earth’s temperature. If ice were to sink, this reflective effect would be reduced, potentially leading to warmer temperatures.
5.3 Geological Impacts
The expansion of water when it freezes can also have geological impacts. When water freezes in cracks and crevices in rocks, it expands and can cause the rocks to break apart. This process, known as ice wedging, is an important factor in weathering and erosion.
6. Investigating Anomalous Expansion: The Full Picture
Water’s anomalous expansion is crucial for understanding its unique properties. Let’s explore the science behind this phenomenon and its implications.
6.1. Temperature of Maximum Density
Liquid water reaches its maximum density at approximately 4°C (39°F). As water cools from higher temperatures, it contracts and becomes denser, just like most liquids. However, below 4°C, water starts to expand, becoming less dense as it approaches the freezing point (0°C or 32°F).
6.2. The Mechanism Behind Expansion
The expansion of water below 4°C is primarily due to the formation of hydrogen bonds. As water cools, these bonds become more stable and organized, creating a structured network. This network forces the water molecules to move slightly further apart, resulting in an increase in volume and a decrease in density.
6.3. Environmental Impact
The fact that ice floats due to this expansion has profound environmental implications.
- Aquatic Life: If ice sank, bodies of water would freeze from the bottom up, killing aquatic organisms and significantly disrupting ecosystems.
- Climate: Floating ice acts as an insulating layer, preventing the underlying water from freezing rapidly. This helps maintain a more stable aquatic environment.
- Geological Processes: The expansion of freezing water can cause weathering and erosion of rocks, shaping landscapes over time.
7. Exploring Supercooling: When Water Defies Expectations
Supercooling is a phenomenon where water remains in a liquid state below its normal freezing point. Let’s examine this intriguing concept and its underlying mechanisms.
7.1. Definition of Supercooling
Supercooling, also known as undercooling, occurs when a liquid is cooled below its freezing point but remains in a liquid state. For pure water at standard atmospheric pressure, the freezing point is 0°C (32°F). However, water can be supercooled to temperatures as low as -40°C (-40°F) under specific conditions.
7.2. Conditions Required for Supercooling
Several factors contribute to supercooling:
- Purity: Highly pure water is more likely to supercool because impurities can act as nucleation sites, initiating ice crystal formation.
- Lack of Nucleation Sites: The absence of surfaces or particles that can act as seeds for ice crystal growth is crucial.
- Rapid Cooling: Quick cooling rates can prevent the formation of ice crystals, allowing water to remain liquid below its freezing point.
7.3. Nucleation and Crystallization
To understand supercooling, it’s essential to know about nucleation and crystallization.
- Nucleation: This is the initial process of forming a new phase (in this case, ice crystals) within a liquid. Nucleation can be homogeneous (occurring spontaneously) or heterogeneous (occurring on a surface or impurity).
- Crystallization: Once nucleation occurs, ice crystals begin to grow. This process involves water molecules attaching to the crystal structure, releasing heat (latent heat of fusion) as they transition from liquid to solid.
7.4. Practical Applications and Examples
Supercooling has several practical applications:
- Cloud Seeding: This weather modification technique involves introducing substances like silver iodide into clouds to act as nucleation sites, promoting ice crystal formation and precipitation.
- Cryopreservation: Supercooling is used to preserve biological materials, such as cells and tissues, at extremely low temperatures to prevent ice crystal damage.
- Instant Ice Packs: Some instant ice packs use supercooled solutions. When activated, the solution rapidly crystallizes, absorbing heat from the surroundings and providing a cooling effect.
8. Molecular Dynamics Simulations: A Microscopic View
Molecular dynamics (MD) simulations provide insights into the behavior of water molecules at the microscopic level, aiding in understanding why ice floats.
8.1. Basics of Molecular Dynamics
Molecular dynamics is a computational method used to simulate the movements and interactions of atoms and molecules over time. By solving Newton’s equations of motion for each atom, MD simulations can predict the behavior of complex systems, such as water, at different temperatures and pressures.
8.2. Modeling Water Behavior
MD simulations of water involve creating a model system with a large number of water molecules and defining the forces between them. These forces typically include:
- Covalent Bonds: Interactions within each water molecule (between oxygen and hydrogen atoms).
- Electrostatic Forces: Interactions between partial charges on different water molecules (hydrogen bonding).
- Van der Waals Forces: Short-range attractive and repulsive forces between molecules.
8.3. Revealing Ice Structure
MD simulations can reveal the structural properties of ice at a molecular level. These simulations show how hydrogen bonds arrange water molecules into a tetrahedral network, forming the characteristic open structure of ice. This open structure is less dense than liquid water, explaining why ice floats.
8.4. Simulating Freezing Process
MD simulations can also simulate the freezing process. As the temperature decreases, water molecules lose kinetic energy, and hydrogen bonds become more stable. These simulations show how water molecules gradually arrange themselves into the crystalline structure of ice, leading to the expansion of volume and decrease in density.
8.5. Advantages and Limitations
MD simulations offer several advantages:
- Microscopic Detail: They provide detailed information about the positions, velocities, and interactions of individual molecules.
- Controlled Conditions: Simulations can be performed under controlled conditions, allowing researchers to isolate specific factors affecting water behavior.
- Predictive Power: MD simulations can predict the properties of water under extreme conditions, such as high pressure or low temperature.
However, MD simulations also have limitations:
- Computational Cost: Simulating large systems for long periods requires significant computational resources.
- Model Accuracy: The accuracy of the simulations depends on the quality of the interatomic potentials used to describe the forces between molecules.
- Complexity: Interpreting the results of MD simulations can be challenging, requiring expertise in computational chemistry and physics.
9. Isotopes of Water: Impact on Density
The isotopic composition of water can also influence its density. Let’s explore how different isotopes of hydrogen and oxygen affect the properties of water.
9.1. Introduction to Isotopes
Isotopes are variants of a chemical element which differ in neutron number, and consequently in nucleon number. All isotopes of a given element have the same number of protons but different numbers of neutrons in each atom.
9.2. Common Isotopes in Water
Water molecules are primarily composed of hydrogen and oxygen atoms. Both elements have several isotopes:
- Hydrogen Isotopes: The most common isotope is protium (¹H), but there are also deuterium (²H or D) and tritium (³H or T).
- Oxygen Isotopes: The most common isotope is ¹⁶O, but there are also ¹⁷O and ¹⁸O.
9.3. Heavy Water
Heavy water (D₂O) is water in which the hydrogen atoms are replaced by deuterium. Deuterium is an isotope of hydrogen with one proton and one neutron in its nucleus, making it about twice as heavy as protium.
9.4. Density Differences
Heavy water is denser than normal water (H₂O) due to the heavier mass of deuterium compared to protium. At 20°C, the density of normal water is about 998 kg/m³, while the density of heavy water is approximately 1106 kg/m³.
9.5. Impact on Freezing Point
The freezing point of heavy water is also slightly different from that of normal water. Heavy water freezes at 3.82°C, which is higher than the freezing point of normal water (0°C).
9.6. Environmental and Scientific Uses
Isotopic variations in water have several environmental and scientific uses:
- Tracers: Isotopes can be used as tracers to study water movement and mixing in hydrological systems.
- Climate Studies: Isotopic ratios in ice cores provide valuable information about past climate conditions.
- Nuclear Reactors: Heavy water is used as a moderator in some nuclear reactors because it is effective at slowing down neutrons without absorbing them.
- Metabolic Studies: Deuterated water is used in metabolic studies to trace the rate of water flux in the body.
10. Practical Demonstrations: Hands-On Experiments
Conducting simple experiments can help illustrate why ice floats on water. These demonstrations provide a tangible understanding of density and buoyancy.
10.1. Simple Density Test
Materials:
- Glass of water
- Ice cubes
- Small objects (e.g., a coin, a piece of wood)
Procedure:
- Fill a glass with water.
- Place an ice cube in the water. Observe that it floats.
- Drop a coin into the water. Observe that it sinks.
- Place a small piece of wood in the water. Observe that it floats (if it’s less dense).
Explanation:
The ice cube floats because it is less dense than water. The coin sinks because it is denser than water. The wood floats if it’s density is less than that of water.
10.2. Freezing Water in a Bottle
Materials:
- Plastic bottle
- Water
- Freezer
Procedure:
- Fill a plastic bottle completely with water.
- Seal the bottle tightly.
- Place the bottle in the freezer and let it freeze.
- Observe the bottle after the water has frozen.
Explanation:
The bottle will likely bulge or crack because water expands when it freezes. This expansion demonstrates that ice is less dense than liquid water and occupies more volume.
10.3. Comparing Ice and Water Levels
Materials:
- Two identical glasses
- Water
- Ice cubes
Procedure:
- Fill one glass with water to a certain level.
- Fill the other glass with the same amount of water.
- Freeze the water in the second glass to form ice.
- Compare the water level in the first glass with the ice level in the second glass.
Explanation:
The ice level will be higher than the water level, demonstrating that ice occupies more volume than the same amount of liquid water.
10.4. Saltwater vs. Freshwater
Materials:
- Two glasses
- Water
- Salt
- Ice cubes
Procedure:
- Fill one glass with freshwater.
- Fill the other glass with saltwater (dissolve salt in water).
- Place ice cubes in both glasses.
- Observe how the ice floats in each glass.
Explanation:
Ice floats higher in saltwater because saltwater is denser than freshwater. The increased density of saltwater provides more buoyant force, causing the ice to float higher.
Have more questions about the fascinating world of science? At WHY.EDU.VN, we’re dedicated to providing clear, accurate, and engaging answers to all your burning questions. Our team of experts is here to help you understand the complex phenomena that shape our world. Visit why.edu.vn today at 101 Curiosity Lane, Answer Town, CA 90210, United States, or contact us via Whatsapp at +1 (213) 555-0101. Let us be your guide to knowledge and discovery.
FAQ About Why Ice Floats on Water
1. Why is water different from other substances in terms of density?
Water’s unique property stems from hydrogen bonding, which causes it to expand upon freezing, making ice less dense than liquid water.
2. Does the size of an ice cube affect whether it floats?
No, the size of the ice cube does not affect whether it floats. The density of the ice compared to the water is the determining factor.
3. Can other liquids float on water?
Yes, any liquid that is less dense than water will float on it, such as oil.
4. Does ice float in all liquids?
No, ice will only float in liquids that are denser than it, such as freshwater or saltwater.
5. What happens if you compress ice?
Compressing ice can cause it to change its crystal structure and become denser, potentially even sinking in water under extreme pressure.
6. How does temperature affect the density of water?
Water is densest at 4°C. Above and below this temperature, its density decreases.
7. Does the salinity of water affect ice’s ability to float?
Yes, ice floats more easily in saltwater because saltwater is denser than freshwater.
8. Are there any practical applications of ice floating?
Yes, it insulates aquatic life, regulates climate, and helps in geological processes like weathering.
9. What is heavy water, and does it affect buoyancy?
Heavy water (D2O) contains deuterium and is denser than normal water, slightly affecting buoyancy.
10. How does supercooling relate to ice formation?
Supercooling is the phenomenon where water remains liquid below its freezing point due to the absence of nucleation sites, affecting ice formation.