Whenever you see blood outside your body, its vibrant red hue is immediately noticeable. But have you ever stopped to wonder, “Why is blood actually red?” The answer lies in fascinating biological and chemical processes happening within our bodies and even across the animal kingdom. Let’s dive into the science behind this vital fluid’s characteristic color.
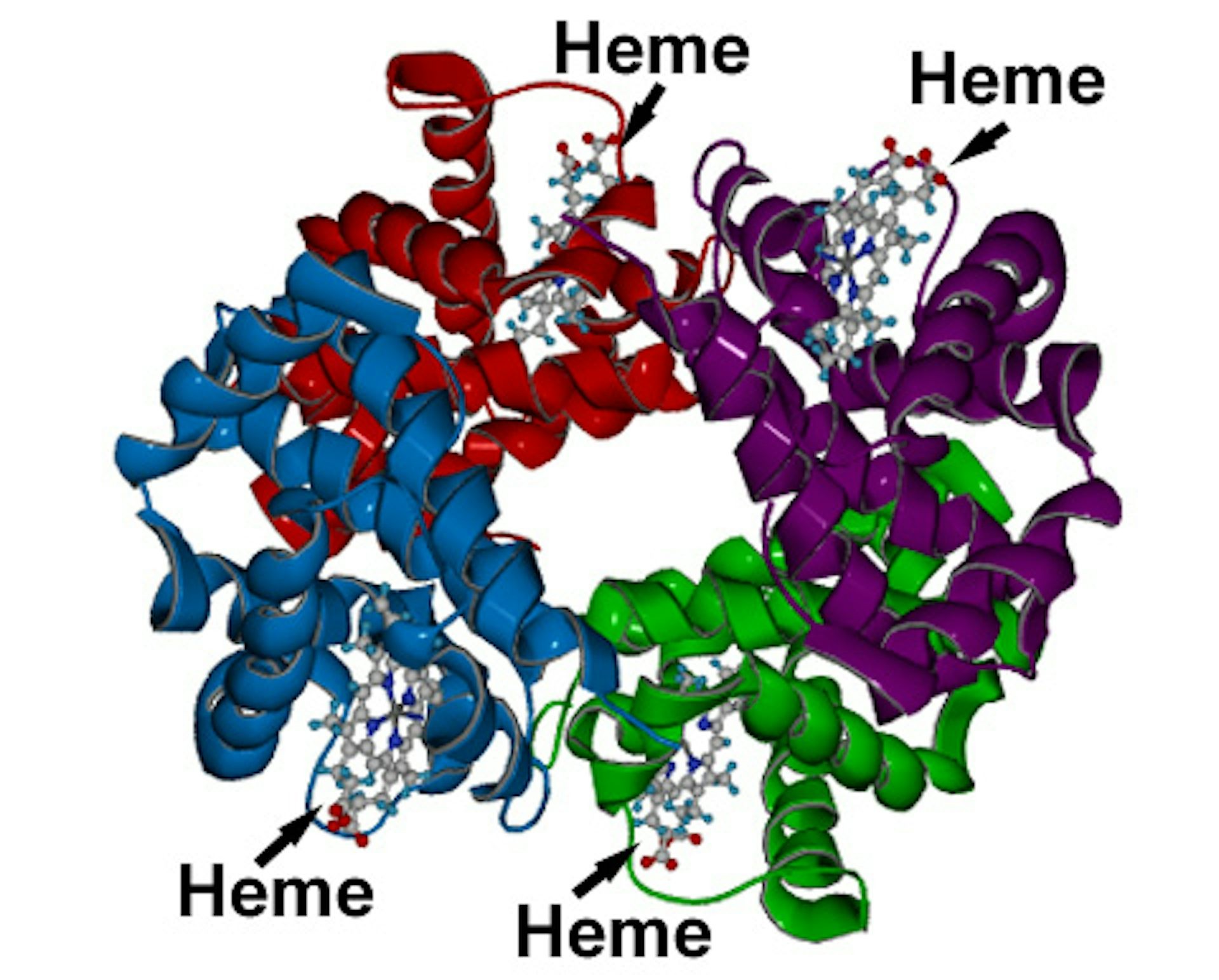
Human blood is red primarily due to a protein called hemoglobin. This protein is packed within red blood cells and is essential for transporting oxygen throughout your body. The key component of hemoglobin responsible for its redness is a compound known as heme. Heme is not just any molecule; it’s a powerhouse containing an iron atom at its center. This iron atom is crucial because it’s the very site where oxygen binds to hemoglobin. Think of heme as the tiny, oxygen-grabbing component within the larger hemoglobin molecule. It’s this interaction with oxygen that sets the stage for blood’s red color.
So, how does this molecular interaction translate to the color we perceive? The secret lies in light absorption and reflection. Everything we see as colored is due to the way substances interact with light. Visible light is composed of a spectrum of colors, each with a different wavelength. When light shines on a substance, certain wavelengths are absorbed, and others are reflected back. Our eyes perceive the reflected wavelengths as color.
Hemoglobin, when bound to oxygen (oxygenated hemoglobin), has a specific way of interacting with light. It absorbs light in the blue-green part of the spectrum. What’s left to be reflected? Predominantly red and orange wavelengths. This reflected light is what reaches our eyes, making oxygenated blood appear bright, cherry red. In contrast, when blood is deoxygenated, meaning it has released its oxygen to the body’s tissues, it absorbs slightly different wavelengths and reflects a darker shade of red, sometimes described as a deep red or burgundy. However, it’s crucial to understand that even deoxygenated human blood is still red, just a different shade.
Interestingly, the color of blood can be dramatically affected by other substances that interact with heme. A prime example is carbon monoxide (CO), a highly dangerous, odorless gas. Carbon monoxide is lethal because it binds to heme at the same iron atom site where oxygen binds, but with a much stronger affinity – about 200 times stronger! When carbon monoxide takes oxygen’s place, it forms carboxyhemoglobin. This not only prevents oxygen from being carried throughout the body, leading to carbon monoxide poisoning, but it also results in a bright cherry-red color of blood. This is why victims of carbon monoxide poisoning can sometimes appear deceptively rosy-cheeked, even in death. The blood remains that bright red because the carbon monoxide stubbornly stays attached to the heme, not allowing oxygen to bind.
A common misconception is that blood becomes blue when it lacks oxygen, especially as seen in veins appearing bluish through the skin. Despite the popular “blue blood” myth, human blood is never blue. The bluish appearance of veins is simply an optical illusion. Blue light has a shorter wavelength and doesn’t penetrate skin and tissue as deeply as red light. When light hits your skin and veins, red light is absorbed more by the blood and surrounding tissues, while blue light is more likely to be reflected back. If a blood vessel, like a vein, is located beneath the skin, more blue light than red light is reflected back to your eyes. This creates the illusion that veins are blue, even though the blood inside is still some shade of red.
While red blood is dominant in many animals, nature showcases a spectrum of blood colors. Blue blood actually exists in creatures like squid and horseshoe crabs. Their blood relies on a different oxygen-carrying molecule called hemocyanin. Instead of iron in heme, hemocyanin uses copper. Copper, when oxygenated, reflects blue light, hence the blue blood. Beyond blue, the animal kingdom exhibits even more surprising blood colors. Some animals boast green, clear, or even purple blood. These variations arise because of different oxygen-carrying molecules in their blood, each interacting with light in its unique way.
Even within red blood, there’s diversity. While most animal blood is red due to hemoglobin, the specific type of hemoglobin can vary across species. These subtle differences in hemoglobin structure allow scientists to distinguish blood samples from different animals, which has applications in fields like forensic science and wildlife biology.
Speaking of forensics, the color of blood can even tell a story over time. When spilled blood dries, the bright red color gradually changes. This is because hemoglobin starts to break down into other compounds, such as methemoglobin, which is darker. As time progresses, further chemical changes lead to the formation of hemichrome, making the dried blood even darker. This continuous color change is not just a curious phenomenon; it’s a valuable tool for forensic scientists. By analyzing the compounds present in a bloodstain and their ratios, they can estimate how long ago the blood was deposited at a crime scene. This can be crucial information in investigations, helping to determine the relevance of blood evidence.
In conclusion, the redness of blood, primarily in humans and many animals, is a fascinating consequence of the interaction between light and hemoglobin, specifically the heme molecule with its iron atom binding to oxygen. While red dominates, the biological world reveals a rainbow of blood colors, each a testament to different oxygen-carrying molecules and the diverse adaptations of life. From understanding basic biology to advancing forensic science, the simple question of “why blood is red” unlocks a wealth of knowledge.